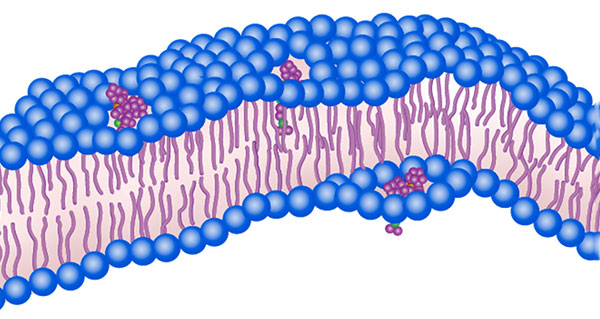
Schematic representation of a molecular machine drilling through the cell membrane as would occur following light activation. [Image: Tour Research Group/Rice University]
To help combat the challenge of antimicrobial resistance, researchers at Rice University, USA, have developed light-activated molecular machines (MMs) that can kill bacteria by drilling tiny holes in their cell membranes (Sci. Adv., doi: 10.1126/sciadv.abm2055). The researchers believe that bacteria would be unlikely to develop resistance against such a mechanical mechanism of action, as compared with a conventional chemical one.
A threat to public health
According to the World Health Organization, antimicrobial resistance is one of the top 10 global public health threats facing humanity today. The phenomenon occurs when bacteria and other microorganisms change over time and no longer respond to medication, leading to more serious infections, longer recovery times and even death.
“New antibacterial agents are urgently needed because bacteria have developed resistance to all or almost all conventional antibiotics,” says Ana Luisa Santos, a postdoctoral fellow and first author of the study. “This means that otherwise treatable infections that could be routinely managed with a course of antibiotics may soon become untreatable.”
To this end, Santos and her colleagues explored a mechanical approach to killing bacteria, which is unlike other approaches involving antibacterial compounds. Their work advances previous research, also at Rice, that demonstrated MMs driven by light that can drill holes in the membranes of individual cells. Notably, in this latest iteration, the nanoscale drills were made to be triggered by visible light (405 nm), rather than harmful ultraviolet light, by adding a nitrogen group.
An effective bacteria-killing machine
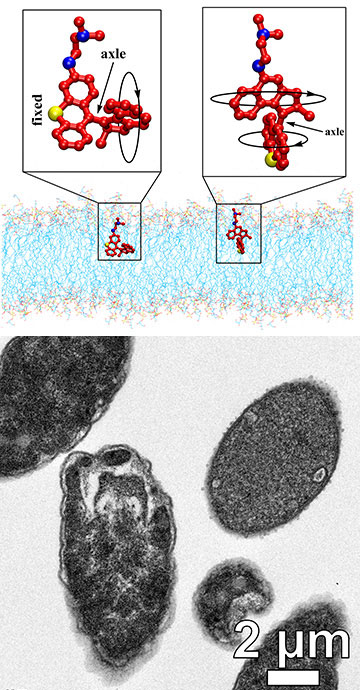
Top: Schematics showing two variants of light-activated molecular machines. Bottom: A transmission electron microscope image showing Escherichia coli bacteria in various stages of degradation after exposure to light-activated molecular drills. [Image: Tour Research Group/M. Meyer/Rice University]
Each MM consists of a paddle-like chain of atoms that acts as a rotor, mounted to a stationary cell-targeting molecule. It binds to the membrane of bacteria, and once activated by LED light, undergoes a conformational change that results in a unidirectional 360-degree rotation at roughly 3 MHz. The MM then drills through the membrane in a matter of minutes, causing the intracellular contents of the bacteria to leak out, ultimately leading to their death.
“In our work, fast molecular machines rapidly killed all strains of bacteria tested, both Gram-negative and Gram-positive, including a methicillin-resistant strain of Staphylococcus aureus (MRSA),” says Santos. “These molecular machines thus have a broad spectrum of antibacterial activity.”
To ensure that the killing was not caused by illumination alone, the researchers performed control experiments under the same conditions without the presence of MMs. They observed only a slight reduction in the number of bacterial cells, whereas the bacterial population was completely eliminated by MMs within as little as two minutes of light activation, which vastly outpaces conventional antibiotics.
Santos and her colleagues also found that repeated exposure to MMs—in this case, 20 cycles—did not result in the development of antibacterial resistance.
Need for precise targeting
For the next generation of nanodrills, the researchers are working on developing MMs that can specifically target pathogens of interest, such as MRSA, in order to minimize damage to host cells and the natural microbiota of the body. Given the need for light to reach the bacteria, they believe that MMs could treat skin infections—in particular, those associated with burn wounds.
“Burn wounds have a high infection rate, and burn wound infection can be a significant cause of death in burn patients,” Santos says. “Molecular machines can be applied locally to the wound and be activated by light to exert their antibacterial activity at the site where it is needed.”

