Feature
Renewed Momentum for Photodynamic Therapy
New approaches are revitalizing photodynamic therapy, a light-driven therapy with more than a century of scientific history.
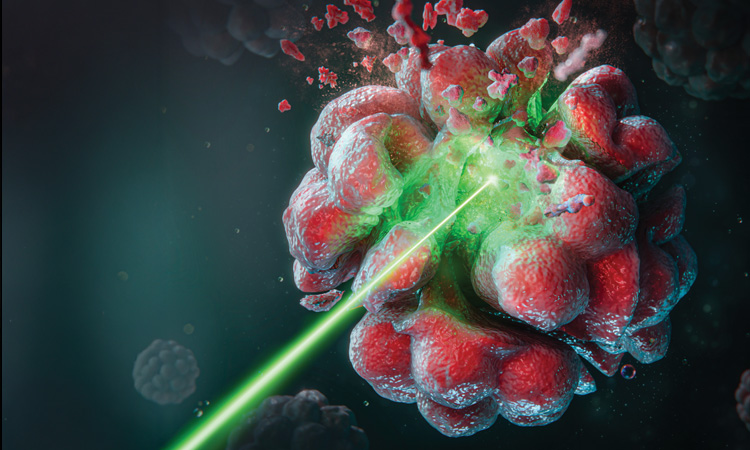 Illustration of single-point laser photodynamic therapy. [C.S. Carrizo et al. Adv. Therap. 8, e00541 (2025)]
Illustration of single-point laser photodynamic therapy. [C.S. Carrizo et al. Adv. Therap. 8, e00541 (2025)]
Since antiquity, humanity has sought to harness the healing power of light. The Ebers Papyrus—a compilation of Egyptian medical texts dated to about 1550 BCE—contains a remedy for vitiligo that combines sun exposure with ingestion of a fruit extract. Hippocrates and other physicians of Ancient Greece promoted the use of sunlight to restore overall health, a practice termed “heliotherapy.” Other civilizations across the world applied sunlight to treat conditions ranging from psoriasis and rickets to skin cancer and psychosis.
…Log in or become a member to view the full text of this article.
This article may be available for purchase via the search at Optica Publishing Group.
Optica Members get the full text of Optics & Photonics News, plus a variety of other member benefits.

