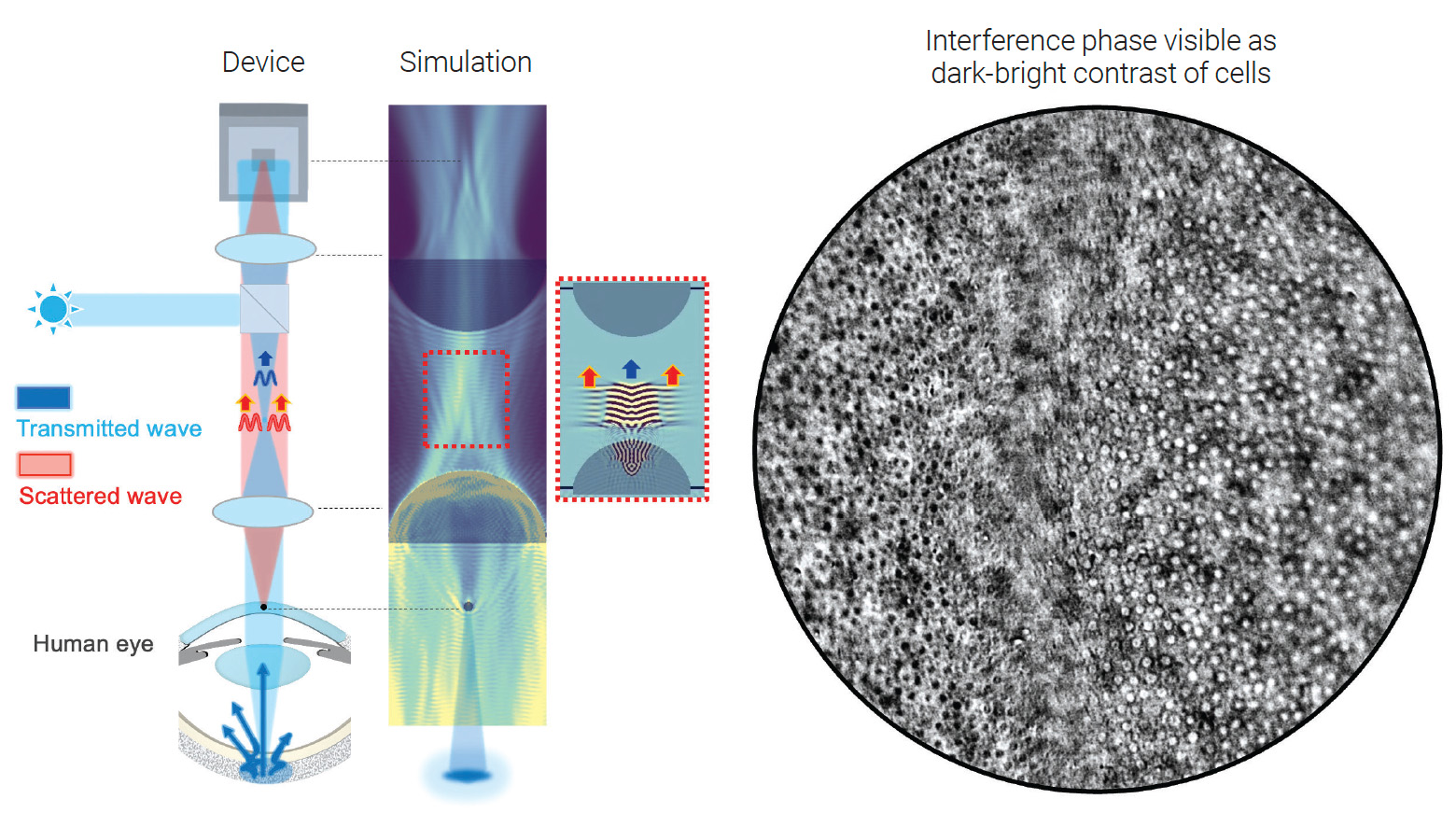
 [Enlarge image]Principle of in vivo transmission interference microscopy, simulation and image of cells, showing interference contrast.
[Enlarge image]Principle of in vivo transmission interference microscopy, simulation and image of cells, showing interference contrast.
Imaging biological tissue in transmission is fundamental to microscopy, yet for a living human eye, it seems impossible—one simply cannot place a light source behind the retina. We have overcome this challenge by reinventing retroillumination microscopy, turning the eye into a high-performance transmission imaging system that reveals cellular structures with unprecedented clarity.1
Our approach is a fundamental advance in retroillumination—the concept of using the back of the eye as an internal light source. While this principle has been used in lower-resolution clinical slit lamps and more recently to achieve phase-gradient contrast microscopy using asymmetric illumination,2 our work is the first to show that it can be leveraged to perform true interference microscopy. Instead of phase-gradient methods, we returned to Ernst Abbe’s foundational theory that image formation in a microscope is an interference phenomenon.3 The final image arises from the interference between the light transmitted directly through the sample and the light scattered by its fine structures.
The key to our innovation is controlling the spatial coherence of the internal light source—the sclera, or white outer layer of the eye. By adjusting the size of the illumination spot projected onto the sclera, one can directly tune the coherence of the retro-illuminated light. A tightly focused spot functions as a small, highly coherent source, analogous to a nearly closed condenser aperture, which dramatically enhances interference contrast. This concept builds on our recent work developing label-free optical transmission tomography based on modulating the Gouy phase shift.4, 5
Our technique enables us to visualize the nearly transparent cellular and nerve layers of the cornea and crystalline lens with remarkable detail across an extended 2-mm field of view. For the first time, the individual crystalline lens epithelial cells, fibers and sutures can be quantitatively analyzed in vivo. Furthermore, in patients with Fuch’s endothelial dystrophy—a major ophthalmic disease affecting 300 million people—transmission contrast highlights the characteristic guttae (excrescences of collagen produced by stressed corneal endothelial cells) with information complementary to that obtained from standard reflection imaging.
Constructed using consumer-grade cameras, the instrument offers a robust, low-cost path for pre-surgical screening and disease monitoring, demonstrating how classical optics can continue to inspire modern solutions to critical health care challenges.
Researchers
Viacheslav Mazlin and Albert Claude Boccara, Institut Langevin, Université PSL, France
Samer Alhaddad, SharpEye SAS, France
References
1. S. Alhaddad et al. Nat. Commun. 16, 7838 (2025).
2. T.D. Weber and J. Mertz, Biomed. Opt. Express 11, 3263 (2020).
3. E. Abbe, Proc. Bristol Naturalists’ Soc. 200, 261 (1876).
4. V. Mazlin et al. Biomed. Opt. Express 13, 4190 (2022).
5. S. Alhaddad et al. Biomed. Opt. Express 14, 4845 (2023).

