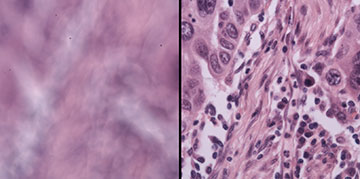
Blurry (left) and in-focus (right) images of human lung cancer tissue before and after DAbI digitally refocuses the image. [Image: Haowen Zhou and Shi Zhao, Caltech]
Every microscope user knows how much time goes into keeping the samples in focus. The constant focus and exposure adjustments can really add up when hundreds or even thousands of specimens need examination.
Now, graduate students at a US university have developed a simple setup for automating the focusing process (Nat. Commun., doi: 10.1038/s41467-026-72287-x ). The technique, based on two LEDs and a physics-based algorithm, also greatly boosts the depth of field of images taken with six types of microscopes.
Previous limitations on autofocus
Moving microscope components to get a specimen into focus is a simple mechanical procedure. Determining the focusing distance between the optics and the sample is the hard part. Conventional microscopes require time-consuming manual adjustments.
Current automated focus methods have disadvantages. Deep-learning methods are only as good as the training datasets that built the model. Passive image-based autofocusing relies on multiple images being captured within a narrow range. Active autofocus techniques require complex hardware. And these challenges only grow when 3D samples are under the microscope.
Two beams of light
Haowen Zhou and Shi Zhao, California Institute of Technology, came up with a simple autofocus technique: shine two closely spaced LED lights, integrated into the microscope, onto the out-of-focus specimen and collect the corresponding images. Adding up the Fourier spectra of the images produces fringes, which correlate to the amount of defocus. Calculating the proper focus becomes a mathematical problem from wave optics.
Zhou, Zhao and their collaborators dubbed the method “digital defocus aberration interference,” or DAbI, and applied it to bright-field imaging, confocal microscopy and several other types of transmission optical microscopes. They also shone the DAbI lights on different samples, both with and without fluorescent labels: human tumor cells, live mouse embryos and cells being screened for defective proteins.
“Our technique, which is enabled by a physics-based observation, is reliable, high performance, and also very simple,” Zhou says. “This makes it useful and powerful for automated, high-throughput microscopy.”
According to the Caltech team, DAbI can keep thin-sample images in focus within a range more than 400 times the depth of focus of a regular optical microscope. For thicker 3D samples—up to 150 μm from top to bottom—DAbI still improves the focus depth by almost 300 times. “This offers truly robust autofocusing of 3D samples, which has never been possible with other techniques,” Zhao says. The Caltech group also says their method works with reflection-type microscopes and could be useful in automated industrial inspections as well as high-throughput biomedical laboratories.

