Prashant Jain led a study that uses solar energy to power a key chemical reaction that drives many manufacturing industries. [Image: Fred Zwicky]
A key step in the manufacturing process for many familiar products—from textiles to detergents—produces harsh byproducts and emits carbon dioxide. Researchers at a US university have developed a plasmon-assisted method to perform the same step with water as the oxidant instead of more hazardous chemicals (J. Am. Chem. Soc., doi:10.1021/jacs.5c18709).
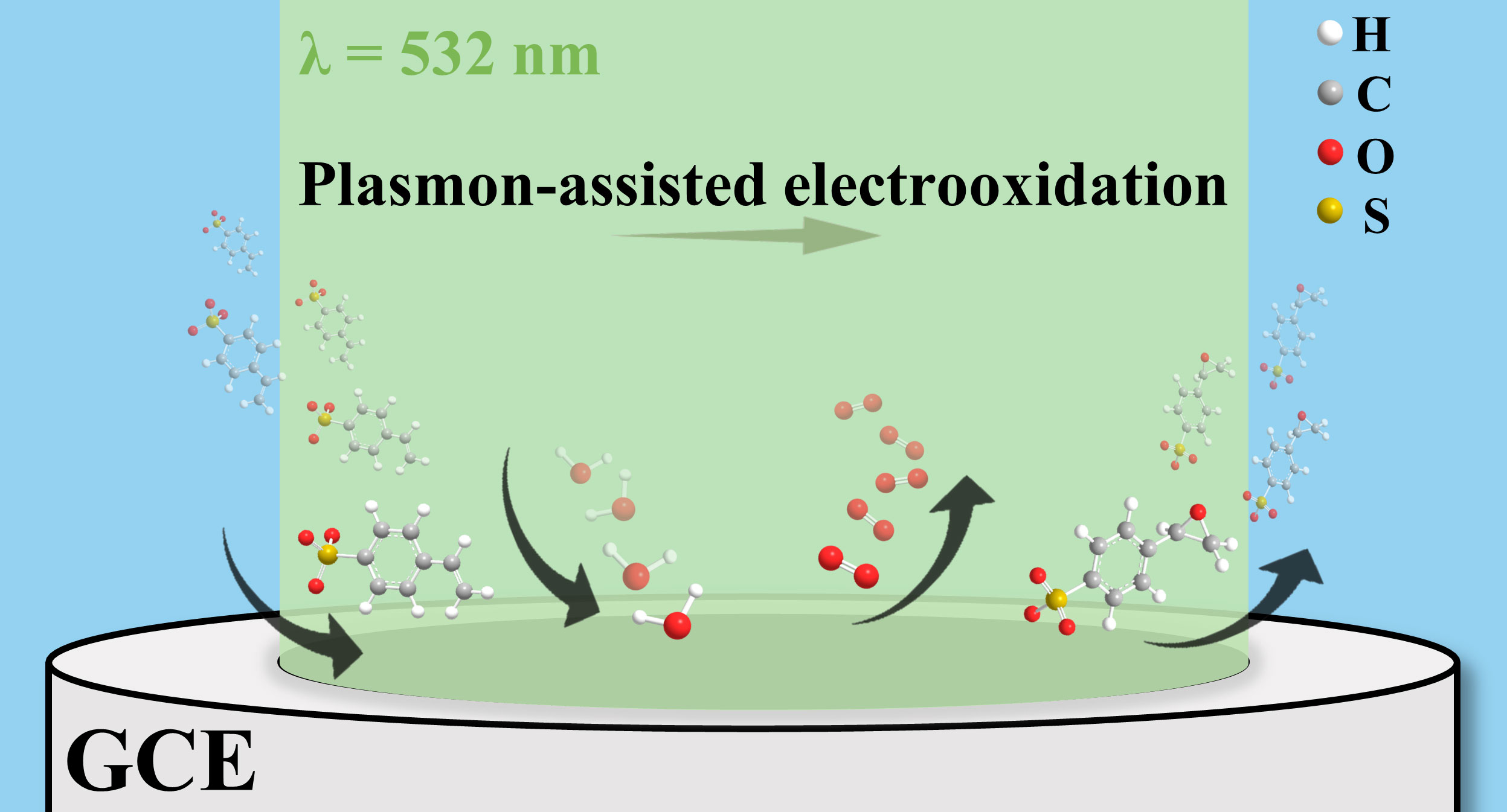
In the presence of plasmonic gold nanoparticles, manganese oxide and water, visible light at 532 nm creates an organic chemical reaction called olefin epoxidation, an intermediate step in the manufacture of plastics and other polymers. The new proof-of-concept process eliminates the need for hazardous peroxides, which release carbon dioxide, and could potentially employ sunlight in place of monochromatic laser light.
Epoxidation with water
In epoxidation, hydrocarbon molecules with a carbon–carbon double bond, known as alkenes or olefins, are converted into epoxides—molecules with ring-shaped structures of two carbon atoms and one oxygen atom. Industrial-scale processes use peroxides or organo-halogen compounds as a source of donor oxygen atoms, but they are hazardous to work with and give off carbon dioxide, one of Earth’s greenhouse gases.
A schematic of plasmon-assisted electrochemical epoxidation of styrene on a glassy carbon electrode (GCE), coated with gold and manganese oxide nanostructures, under the illumination of a 532-nm laser light source. In this graphic, H = hydrogen, C = carbon, O = oxygen and S = sulfonate from styrene oxidation. [Image: Courtesy of Prashant Jain]
Using water as an oxidant in epoxidation would be safer for both the chemical factory and the planet. However, the bonds in H2O require high amounts of energy to break, and the required heat would lead to further greenhouse emissions.
Boosting electrochemistry with lasers
A team led by chemist Prashant Jain of the University of Illinois Urbana-Champaign, USA, knew that visible light can excite localized surface plasmon resonances on metallic nanostructures, thus intensifying electrocatalytic reactions. Previous work had applied the plasmonic technique to ammonia synthesis, so Jain’s group investigated whether the method could apply to other industrial chemicals.
Jain’s colleague Lucas Germano created tiny light receptors made of manganese oxide rods coated with gold nanoparticles. Under a transmission electron microscope, these structures look like spotted needles less than 1 μm in length. The scientists measured the amounts of the initial reagent (4-styrenesulfonate), epoxidation product (styrene oxide sulfonate) and other byproducts in the presence of the catalyst structures, both with and without laser irradiation. The plasmon excitation due to the visible light created five times more epoxidation product than experiments conducted in darkness.
According to the research team, gold nanoparticles are much more stable in the electrochemical oxidation process than silver or copper, and gold’s interband transitions under visible light conditions excite plasmons into generating high-energy holes. The scientists hope to apply this method to other electrochemical processes that could use water as a safer oxidant—and perhaps make the technique even “greener” with sunlight instead of laser light.