![]()
An artist’s imagining of an intricate internal microfluidic porous structure, made through the interaction of photosensitive polymers and micro-LED light in a process designed by a team at the Kyoto University Institute for Integrated Cell-Material Sciences. [Image: Andrew H. Gibbons / Kyoto University iCeMS]
Researchers in Japan have developed a photolithography technique called organized microfibrillation (OM) that can create complex, self-enclosed channels within a polymer film (Nat. Commun., doi: 10.1038/s41467-022-29956-4). According to the team, OM has a number of advantages over traditional methods for fabricating microfluidic devices, which are currently employed in DNA sequencing, point-of-care diagnostics and organ-on-a-chip models.
“This is an easy technique for making microfluidic channels,” said study author Andrew Gibbons of the Kyoto University Institute for Integrated Cell-Material Sciences (iCeMS). “The porous structure is made directly in film, so no assembly is required. And it doesn’t require making an etched silicon mold first, so designs can be changed as required.”
From carbon capture to microfluidics
Gibbons and his colleagues belong to iCeMS’s “Pureosity” team, whose research focuses on carbon-capture membrane technology. In their quest for novel ways to produce materials with controlled pore sizes, they decided to experiment with photolithography, drawn by the flexibility and precision that light sources could potentially afford.
After first demonstrating the technique in 2019, the researchers realized that OM could be harnessed as a user-friendly, high-resolution technology platform for printing microfluidics. The current study delves into that application with a proof-of-concept microfluidic OM device that can successfully separate biomolecules by size.
“Microfluidics is an exciting field with a lot of potential, so we were keen to push the research in this direction,” said Gibbons. “An application we have in mind is wearable sensors, where sweat can be drawn into the device and tested locally for specific biomarkers.”
Structural-color effects
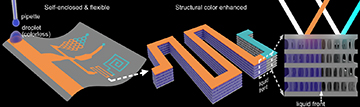
In the proof-of-concept, OM microfluidic channels were fabricated by first illuminating a photosensitive polymer film on a reflecting surface with monochromatic light. The reflection of light creates a standing-wave interference pattern in the polymer film, which determines which regions become cross-linked by the light. A shadow mask was used to create the desired patterns. The researchers note in the paper that they used “a micro-LED illumination instrument” for the cross-linking, which enabled control of illumination down to the 1-µm level.
The periodic structure created by the iCeMS team’s technique gives rise to structural-color effects in the channels when illuminated by incident light—with the color changing with the onset of flow in the channel, and with variations in the flow rate. [Image: D. Qin et al., Nat. Commun., doi: 10.1038/s41467-022-29956-4 (2022); CC-BY 4.0] [Enlarge image]
Then, the film was developed in a weak solvent that breaks down the polymer in any non-cross-linked regions. The finished product contains self-enclosed channels with a unique internal porous structure, composed of periodic non-porous layers separated by porous microfibril layers. The Bragg reflections of incident light due to the alternating layer structure create structural-color effects. And, as the team experimented with the device, they found that the color of the film correlated with pore size and liquid flow rate in the microchannel—a useful property for visualizing the flow properties of prototype devices.
In addition, controlling the cross-linking energy allows the creation of varying porosity at submicron scale in a single miniature device. As a demonstration, Gibbons and his colleagues created a miniature OM device that could separate two proteins of different molecular weights.
“We are working on how to make printing with our technique more practical, and we are looking at how different biomolecules can be separated within our films for diagnostic purposes,” he said. “We are excited to see where we will go next with our new technique.”