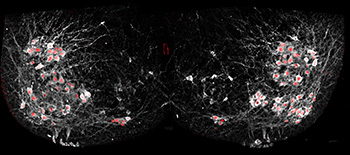
Salk scientists selectively added fluorescent proteins motor neurons (red) to show how cells in the spinal cord synchronize many neurons at once to allow complex movements. Credit: Christopher Hinckley / Pfaff Lab, Salk Institute
Researchers from the Salk Institute for Biological Studies (USA) have developed a method of imaging the intraspinal activity of motor neurons in real time (Neuron, doi: 10.1016/j.neuron.2015.08.005). Using mouse models with genetically altered motor neurons that fluoresce when activated, they used two-photon imaging to view the activity of several neurons at once—without any post-image processing. Their study results showed that the spinal neurons didn’t always deliver “instructions” from the brain to motor neurons based solely on their location, but that the genetic subtype of the motor neuron was also sometimes important. The authors say this information suggests that treatment with specific types of motor neurons, not general motor neurons, may be the best treatment for neurodegenerative diseases.
The locomotor central pattern generator (CPG) is a group of cells in the spinal cord that sends action messages to motor neurons that control body movement. For example, the CPG delivers the brain’s instructions to the motor nerves responsible for the muscles involved with walking. Previous methods for observing neuron activation involved electrodes—but they only allowed for the observation of one cell at a time. And, until now, researchers have been unable to determine how and if motor neuron position and genetics play a role in the coordination of complex motor behaviors.
To observe the connection between neurons in the CPG and motor neurons, the group from Salk, led by Samuel Pfaff, tagged mouse motor neurons with GCaMP6f—a protein that lights up when the neurons are activated. They viewed in real time the patterns of CPG and various types of motor neuron activation in vitro with a microscope. (A video of the activation patterns can be seen here.) The imaging was conducted at 8.3 frames per second with a field of view of 550 x 550 µm. The Salk team discovered that the location of the motor neuron is not the only way a CPG identifies the recipient of its message; the motor neuron’s genetic subtype sometimes plays a role in firing patterns.
