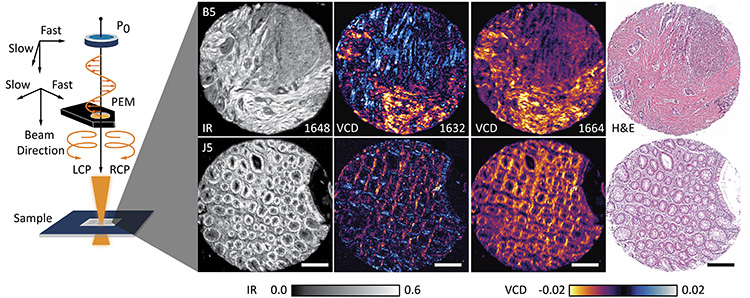
 Left: Schematic showing generation of left and right circularly polarized light states in the infrared (IR) using a photoelastic modulator. Right: IR absorbance images, two recorded VCD images, and histologically stained images of two cores from a tissue microarray with normal (J5; bottom row) and cancer-bearing (B5; top row) colon tissue. Scale bars: 250 µm.
Left: Schematic showing generation of left and right circularly polarized light states in the infrared (IR) using a photoelastic modulator. Right: IR absorbance images, two recorded VCD images, and histologically stained images of two cores from a tissue microarray with normal (J5; bottom row) and cancer-bearing (B5; top row) colon tissue. Scale bars: 250 µm.
Chirality is observed across all biological systems, providing a specificity wherein the molecules of life occur in non-superimposable mirror images of each other. For instance, amino acids are typically in L form, whereas sugars are in D form. Molecular aggregation that provides functional units in organisms can also result in supramolecular chirality. Since all biological processes are chiral-specific, chirality mapping is crucial in studying development and disease processes.
However, there is still no viable tool for spatial-specific mapping of molecular chirality. Chiroptical techniques such as Fourier transform-infrared (FT-IR) spectroscopy can measure vibrational circular dichroism (VCD) and quantify the differential response of molecules to circularly polarized light.1 But such techniques typically require hours for spectral acquisition and are limited to samples in liquid state. Extending them to imaging is challenging since the signal at each pixel is approximately 104 times lower than absorbance measured in bulk spectrometers.
We developed a novel, widely tunable quantum cascade laser–based design.2,3 Instead of acquiring entire spectra through interferometry, we employed a discrete-frequency approach that utilizes the intrinsic polarization of the laser source. We modified a confocal laser scanning microscope4 with a photoelastic modulator, and validated it for VCD measurements.5 While absorption of mid-IR light provides information about the sample’s molecular composition at each pixel, the differential absorption of left and right circularly polarized light can potentially quantify molecular chirality.
Our work addresses two major bottlenecks—a limited signal-to-noise ratio (SNR) and long acquisition times—that have made FT-IR VCD instruments impractical for imaging. Being time intensive, VCD measurements cannot be used to image complex biological systems or solid tissue. Current commercial VCD instruments take ~hours to acquire a single VCD spectrum. Our system’s better SNR allowed reliable acquisition of VCD signals at the micron-pixel level. We acquired VCD spectra of different protein samples and assessed their secondary structures in minutes.
Finally, we explored the ability to detect VCD signals from tissue sections, providing new opportunities for optical modeling to understand the origin of linear and circular dichroism and absorbance signals while relating them to underlying molecular processes. Our work provides a tool for future studies of small, localized changes in tissue that have implications for systemic diseases and their progression. Since chirality is fundamental to life itself, we believe that this technique could help uncover intricate biological processes, organization in tissues and drug therapeutics.
Researchers
Yamuna Phal, Kevin Yeh and Rohit Bhargava, University of Illinois at Urbana-Champaign, Urbana, IL, USA
References
1. L.A. Nafie. Chirality 32, 667 (2020).
2. K. Yeh et al. Anal. Chem. 91, 2177 (2019).
3. Y.D. Phal et al. Appl. Spectrosc. 75, 1067 (2021).
4. S. Mittal and K. Yeh et al. Proc. Natl. Acad. Sci. USA 115, E5651 (2018).
5. Y.D. Phal et al. Anal. Chem. 93, 1294 (2020).

