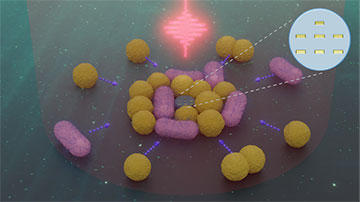
An artist’s impression showing how a nanoscale robot (center) containing seven gold rods can attract bacteria to it via the thermophoretic force while being illuminated by a laser. [Image: Jin Qin / Uni Würzburg]
Scientists in Germany have used laser light to guide micro- and nano-scale robots along complicated trajectories at high speeds (Nat. Commun., doi: 10.1038/s41467-026-70685-9). By exploiting local optical and thermal forces, they also showed how these robots can transport bacteria from one point within an aqueous solution to another, which they say suggests a new kind of robotic cleaner for use in microbiology and biomedicine.
From two lasers to one
The work, carried out by Bert Hecht and Jin Qin, University of Würzburg, and colleagues, relies on the thrust and torque generated when light interacts with tiny optically resonant structures. Previously, the researchers demonstrated “microdrones” featuring four plasmonic antennas that each scatter light in specific directions. They showed how to control the devices’ translational and rotational movement in two dimensions—despite the influence of Brownian motion—by varying the intensity and polarization of a pair of laser beams.
They have now obtained similar results from a simpler system, using interactions with a single laser beam to both propel and maintain the trajectory of tiny robots. They made their devices by carefully arranging gold rods with varying lengths, slightly greater than 100 nanometers, inside a transparent silica disk.
Tiny, steerable robots
The researchers found that after releasing the robots into an aqueous solution they could guide them around a rectangular circuit at several tens of micrometers a second.
The researchers began by making a 1.5-µm-diameter “microrobot” comprising four pairs of gold rods arranged in a cross formation. The upper and lower pairs served as thrusters, scattering laser light asymmetrically such that it interfered constructively on one side of the rods’ axes and destructively on the other. The pairs of rods on either side of the structure instead served to steady the robot’s course, generating a corrective torque whenever the rods became unaligned with the laser’s polarization axis.
They followed this up with a slightly smaller “nanorobot,” featuring a more compact arrangement of antennas to perform the same functions. This measured just a fraction under 1 µm across, while weighing a mere 0.26 trillionths of a gram.
In both cases, the researchers found that after releasing the robots into an aqueous solution they could guide them around a rectangular circuit at several tens of micrometers a second—following one set of parallel sides by shining a vertically polarized beam at the tiny devices, while using horizontally polarized light for the other set. They say that it would in principle be possible to negotiate the corners simply by switching between the two linear polarizations, but that in practice they need to impart a short pulse of circularly polarized light to avoid the disk’s inertia causing the robot to turn the wrong way.
Trapping bacteria with nanorobots
With this basic demonstration under their belt, Hecht and colleagues went on to feed a series of preprogrammed voltage waveforms to electro-optical modulators such that the resulting sequence of laser polarizations sent the nanorobot along more complex trajectories, including that of the characters “EP5.” (They note that the lines traced out by the robot were a little jagged, due to a minimum level of Brownian motion and still imperfect cornering.)
They then showed how the robots can be used to marshal bacteria in solution, thanks to the trapping effects of both the nanostructures’ optical near fields and local heating (causing a photothermal effect). They found that both the larger and smaller devices could drag clusters of Escherichia coli and Staphylococcus carnosus through the liquid, picking them up with the laser turned on and then dumping them once the light was turned off. Remarkably, they say, the robots were able to maneuver smoothly even when carrying bacterial clusters weighing hundreds of times their own weight.
According to the researchers, the new system could function as a “nanoscale biological cleaner,” collecting bacteria in one region and then placing them in another without damaging them. They reckon that the devices should be versatile and precise enough to support applications as varied as bioengineering, targeted drug delivery and nanoscale sensing.
The scientists claim that the system has a number of advantages over conventional optical tweezers, including the need for much lower laser intensities and less heating of bacteria. What’s more, they say that the ability to transport multiple bacteria simultaneously—in contrast to the single units captured by optical tweezers—should significantly increase throughput.
Looking ahead, Hecht and colleagues say it should be possible to operate several robots at the same time by using a device such as a spatial light modulator to divide a single laser beam into multiple subfields, each with its own polarization state and intensity profile. They say that should facilitate additional operations, like squeezing bacteria, or, with the addition of laser-driven heating, slicing open cells. But doing so, they add, will require new hardware to provide a suitably powerful and stable beam as well as the necessary high-bandwidth control.

