Illustration of a colon cancer cell. [Image: Kateryna Kon / Science Photo Library / Getty Images]
The treatment of malignant tumors has long presented a challenge for cancer researchers. Bacteria-based therapies—in which microbes, often souped-up by genetic engineering or nanotechnology, are put to work as on-site cancer killers—show promise for improving upon conventional methods. But these techniques have drawbacks, including the risk of antibiotic resistance and the need for complicated procedures that could degrade the bacteria.
Now, researchers in Japan have demonstrated a new approach for enhancing purple photosynthetic bacteria, which they say is an ideal strain for effective cancer phototherapy (Nano Today, doi: 10.1016/j.nantod.2023.101966). Their process, which involves simple chemical functionalization, preserves the innate medicinal qualities of the bacteria while enhancing their ability to fight cancer.
Picking purple bacteria
The researchers chose the purple photosynthetic bacteria Rhodopseudomonas palustris (RP) as an optimal candidate for cancer treatment because it is spatiotemporally activatable by near-infrared light and shows strong photothermal conversion—the ability to turn laser light energy into heat, in this case to selectively eliminate cancer cells. This is thanks to its bacteriochlorophyll (BChl) light-harvesting nanocomplexes, which are useful for targeted optical cancer therapies.
“RP demonstrated excellent properties, such as near-infrared (NIR) fluorescence, photothermal conversion and low cytotoxicity,” explained lead author Eijiro Miyako, Japan Advanced Institute of Science and Technology (JAIST), in a press release accompanying the paper. “It absorbs NIR light and produces free radicals—a property that can be utilized to kill cancer cells.”
Chemical functionalization
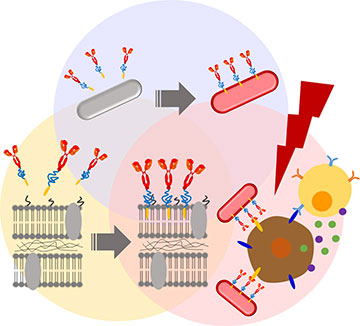
The membranes of photosynthetic bacteria were PEGylated, and fluorescent markers and an anti-PD-L1 antibody were attached to enable tumor-targeting and immunological activation. The engineered bacteria demonstrated effective tumor suppression and immunological responses in a mouse model of colon cancer. [Image: Eijiro Miyako] [Enlarge image]
After selecting their preferred bacteria, the researchers looked to improve them through a series of modifications. First, they attached polyethylene glycol (PEG) derivatives, including one called Biocompatible Anchor for Membrane (BAM), to the bacterial cell walls. Known as PEGylation, this process has a number of benefits, including helping the complex evade host immune response and facilitating attachment of other biomolecules. The team then affixed a fluorescent Alexa488-BSA conjugate to the BAM, which allowed it to be tracked with fluorescence microscopy and used for locating tumors.
Finally, the researchers tacked on an immune checkpoint inhibitor antibody known as anti-PD-L1 using the same BAM method. Cancer cells express a protein called Programmed Cell Death Ligand 1 (PD-L1), which suppresses the host’s immune response and allows cancer cells to evade detection and elimination. Anti-PD-L1 antibodies block PD-L1, thus preventing cancer cells from flying under the immune radar and allowing them to be targeted by the host’s immune system.
Getting a laser boost
To examine the efficacy of the various bacterial complexes, the researchers pitted them against colon cancer in mice in a series of experiments. Tests showed that anti-PD-L1–BAM–RP, BAM–RP and RP inhibited tumor growth when injected in mice with colon cancer. However, all the varieties had an especially dramatic anticancer effect when excited with an NIR laser at 0.7 W for 3 minutes.
During the 30-day follow-up period after the experiment, solid tumors disappeared completely in mice that underwent laser irradiation of injected anti-PD-L1–BAM–RP, BAM–RP, or RP. Laser-induced anti-PD-L1–BAM–RP was the most effective during the primary treatment stage and also cured tumors faster than the others.
“Our findings revealed that light-driven functional bacteria demonstrated effective optical and immunological functions in the murine model of colon cancer. Moreover, the NIR fluorescence of the engineered bacterial complexes was used to locate tumors, effectively paving the way for future clinical translation,” says Miyako. “We believe that this bacterial technology could be available for clinical trials in 10 years and have positive implications for cancer diagnosis and therapy.”