
In 2007, the Beijing Olympic Stadium, colloquially known as the “Bird’s Nest,” appeared surrounded in smog and haze. [Ry Tweedie-Cullen]
From the world’s most developed cities to its least developed rural areas, air quality is in the news like never before. Concerns over climate change, leaks from hydraulic fracturing (“fracking”) sites, adverse effects of air pollution on public health and the potential for explosions from aging natural-gas pipes all drive the demand for rapid, portable detection systems for trace gases.
Mid-infrared laser diodes can now provide light for absorption spectroscopy in a variety of configurations. The most important members of this group are quantum cascade lasers (QCLs), which over the past two decades have evolved from being new discoveries in the optics laboratory to robust, commercially available units.
Now that these devices are on the market, environmental scientists have been devising schemes to take mid-infrared sensing out of the laboratory and onto the rooftops, streets and drilling sites where trace gases raise potential problems. Combined with the proper reflectors and detectors, the QCL-based systems can spot-check for natural-gas leaks before they can blow up buildings and provide ground-based verification of remote sensors on aircraft and spacecraft.
“Chemical sensing relies very heavily these days on optical detection, and if you can target the most intense light-matter interactions, then you can build better sensing systems,” says Gerard Wysocki, an electrical engineering professor at Princeton University (New Jersey, USA).
Much of the research has been taking place under the aegis of the Center for Mid-Infrared Technologies for Health and the Environment (MIRTHE), based at Princeton University and Rice University (USA) in collaboration with four other U.S. universities. Among other research campaigns, scientists at the federally funded center have brought QCLs to at least three continents to examine the air inhaled by Olympic athletes and rural villagers.
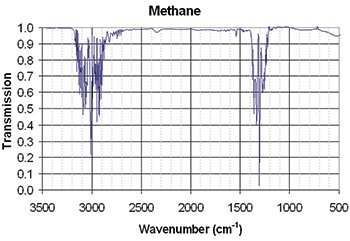 Infrared spectrum of methane, showing features in the regions of wavenumber 3,000 and 1,300 cm–1 (λ=3.3 and 7.7 μm). [Wikimedia Commons]
Infrared spectrum of methane, showing features in the regions of wavenumber 3,000 and 1,300 cm–1 (λ=3.3 and 7.7 μm). [Wikimedia Commons]
Properties of QCLs
Almost as soon as QCLs were invented in 1994, scientists realized that they could play an important role in mid-infrared spectroscopy. Prior to the advent of QCLs, researchers did not have laser sources that could span the two windows, 3 – 5 μm and 8 – 13 μm, where the atmosphere is most transparent to water vapor.
Many trace gases in the atmosphere, including methane, have strong absorption features in the mid-infrared spectral region at which QCLs operate, says Anna Michel, an assistant scientist at Woods Hole Oceanographic Institution (USA). The mid-infrared is the so-called “molecular fingerprint” region where all organic and many inorganic molecules exhibit fundamental rotational and vibrational resonances.
Mid-infrared spectroscopy requires a much shorter path length than near-infrared spectroscopy to yield the same level of sensitivity, says Princeton environmental engineering professor Mark A. Zondlo, who leads the university’s atmospheric chemistry research group and also serves on the MIRTHE faculty. The Beer-Lambert law permits the mid-infrared instrument to be more compact, requiring less power to operate and with less mass to tote around—true portability for a variety of situations.
 Harvard graduate student Ben Lee tunes the optics on a NASA QCL methane isotope sensor. [NASA Dryden / Tom Tschida]
Harvard graduate student Ben Lee tunes the optics on a NASA QCL methane isotope sensor. [NASA Dryden / Tom Tschida]
Zondlo says he has experimented with QCLs for a decade, but only in the past five years or so have the commercially available QCLs advanced enough in terms of beam quality, thermal stability, efficiency and general ruggedness to take their place in field spectroscopy campaigns to answer new science questions.
The concept of making integrated measurements of gases along a line of sight is not new, according to electrical engineering professor Fred Moshary of the City College of New York (CCNY; USA): Fourier transform infrared spectrometers already make short-path air-quality measurements at various locations around cities, particularly for homeland security. However, QCLs can potentially reach much higher resolutions and much faster response times, and accomplish that with simpler, more robust equipment.
Quantum cascade and interband cascade lasers
In 1994, OSA Fellow Federico Capasso, Jérôme Faist and their colleagues at AT&T Bell Laboratories (USA) invented a semiconductor laser that worked differently from all other laser diodes that preceded it. Their unipolar device generated light via intersubband transitions in a stack of quantum well heterostructures, so the inventors dubbed it the quantum cascade laser (QCL).
The first QCL was suited only for laboratory use, as its highest operating temperature was a frosty 125 K. It gave off 4.3-μm-wavelength radiation in pulsed mode with a peak power of 30 mW. Rapid innovation in the field led to room-temperature operation, CW operation and the development of several laser types: Fabry-Pérot, distributed feedback and external cavity.
Interband cascade lasers, based on slightly different physics, were developed by groups at several U.S. and Canadian institutions in the mid-1990s. These lasers generate photons through interband transitions, rather than intersubband transitions.
QCLs were commercialized in 2004, and interband cascade lasers soon followed them into the marketplace. Today’s researchers can focus their attention on developing room-temperature applications for these compact lasers, instead of the lasers themselves.
Laser absorption spectroscopy at the 2008 Olympics
As China prepared to host the 2008 Summer Olympics in Beijing, critics expressed concern that the bad air of the megalopolis could be detrimental to the athletes. [A 2015 article in PLoS ONE reports that that air pollution contributes to as many as 1.6 million deaths per year in China.] Shortly before the Olympics began, Chinese officials took steps to improve the city’s air quality: halting construction, putting severe restrictions on driving, and promoting public transportation. Several MIRTHE graduate students were building computer models to predict the air-quality conditions during the Games; members of the Chinese Academy of Sciences were collaborating on the modeling work and also measuring ozone at a site close to the Beijing National Stadium—nicknamed the “Bird’s Nest”—and the National Aquatics Center (the “Water Cube”).
Michel, then a MIRTHE postdoctoral fellow at Princeton, and her colleagues brought a novel sensor they dubbed the Quantum Cascade Laser Open-Path System (QCLOPS) to Beijing to detect trace amounts of gases in the city’s air before, during and after the 2008 Olympics. The open-path sensor system (sometimes called a long-path or fence-line sensor) consists of a pulsed QCL as a transmitter, a retroreflector to reverse the beam’s direction and a telescope-style receiver mounted alongside the transmitter. Between the transmitter/receiver and the reflector lie many meters of open air. (In some open-path setups, the transmitter and receiver are on opposite ends of the beam path.) “The idea is that if you can build these sensors that cover a very long distance, then you can get an integrated measurement,” Michel says.
A fixed-point sensor, measuring at only one location, may be near a “hot spot” of high trace-gas emission or at a place where none of the gas of interest exists, according to Michel. On the other hand, a long-path or open-path sensor provides an integrated measurement over the whole distance between the laser and the receiver. “So you’re going to get a much better representation of your sample than if you just took one measurement in one location,” Michel says.
Michel’s team set up its instruments on the roof of a Chinese Academy of Sciences building in downtown Beijing, about 3.2 km from the Bird’s Nest. The laser and receiver—a Newtonian telescope coupled to a cooled mercury cadmium telluride detector—sat in an enclosed room on the roof and pointed out the window to a retroreflector at the opposite end of the roof. The beam’s total round-trip path length was 76 m, limited by the size of the building.
Supplying the QCLOPS beam was a commercially available external-cavity QCL, broadly tunable from 9.3 to 9.8 μm. That region includes absorption lines for ozone, carbon dioxide and ammonia. The laser’s wide tunability, due to the external cavity, allowed the researchers to target multiple chemical species, such as ozone and carbon dioxide, with only one laser. In future environmental applications, Michel says, such tunability could discriminate between isotopes of carbon, hydrogen and oxygen, which could yield clues to the origins of the gases.
QCLOPS operated 24 hours a day for 10 weeks with, as the team wrote in a journal article, “minimal human intervention.” The system stopped collecting data during rain showers, but resumed once the retroreflector was dry.
Michel and the MIRTHE team compared their data with those gathered by a commercial point sensor provided by their Chinese colleagues. Both systems captured the diurnal changes in ozone levels between 0 and 1,500 parts per billion by volume (ppbv). [One report from the University of Rochester (USA) estimates that the government restrictions made concentrations of different air pollutants fall by up to 59 percent.]
By running what she now considers a first-generation system in Beijing, Michel says, “We not only learned a lot from an environmental standpoint, but also from a design standpoint—how to improve upon these sensors.”
 Integrated open-path sensing at Toolik Lake, Alaska, USA. (Inset) The optical instruments on the lab bench. [Anna Michel]
Integrated open-path sensing at Toolik Lake, Alaska, USA. (Inset) The optical instruments on the lab bench. [Anna Michel]
Improving open-path sensors
Over the next few years, Michel worked with Zondlo and David Miller (now a postdoc at Brown University, USA) to study the Arctic’s changing levels of methane. “Methane is 25 times more potent a greenhouse gas than carbon dioxide on a per-molecule basis,” points out Zondlo. Thus, he says, if natural-gas vehicles have large methane leaks, they can have a disproportionate impact on climate.
Levels of this trace gas fluctuate for various reasons. For example, methane is released when permafrost thaws, or it may be trapped under the ice of a frozen lake and released when the ice melts. Methane could also leak from a gas pipeline or a fracking site. Such transient emissions are challenging for environmental scientists to study; although they have a pretty good handle on the overall methane budget of the atmosphere (i.e., all the methane entering and leaving the atmosphere), they have a harder time pinning down quantities of individual methane sources.
In 2012, Michel, Zondlo and Miller were part of a team that spent two weeks at the University of Alaska’s Toolik Field Station, which sits on the shore of a lake on Alaska’s North Slope, a five-hour drive south of the Arctic Ocean oil port of Prudhoe Bay. On opposite sides of an inlet on Toolik Lake, the scientists deployed a retroreflector opposite a CW, 8-μm distributed-feedback QCL with 40 mW of output power. They repositioned the equipment multiple times over the two weeks of observations, so they ultimately collected their open-path measurements over round-trip path lengths of 0.45, 0.75 and 1.2 km.
One of the biggest challenges the group faced was aligning the laser/detector and the retroreflector given the long hours of daylight above the Arctic Circle in August—the scientists had about half an hour of dusk to use a green laser to check the alignment of the components, Michel says. Despite the obstacles, as the team reported to the 2013 CLEO conference, the long-path sensor picked up the spectral signature of methane, maintained a stable alignment and kept operating through high winds, rain and even snow.
Since Toolik Field Station is a permanent facility that hosts visiting scientists in a variety of disciplines, it provides electrical power. However, Michel wants her next field-portable sensing instrument to run entirely on batteries or solar panels. Her group at Woods Hole has received funding from a private foundation to build a third-generation long-path system using an array of 32 QCLs. Having so many lasers tuned to slightly different frequencies will allow researchers to target more than one gas at a time, Michel says, and to use spectral fitting techniques to improve overall sensitivity. The group currently has funding to test the system at Woods Hole, but not enough funding for testing at a remote location.
 Open-path laser measurements across CCNY campus. [Paulo Castillo / Brookhaven National Laboratory]
Open-path laser measurements across CCNY campus. [Paulo Castillo / Brookhaven National Laboratory]
Hitting the road with open-path systems
Open-path sensors have been tested in different situations around the world. For example, in 2010, a MIRTHE-affiliated Princeton graduate student, Ekua Bentil, brought QCLOPS to her native country, Ghana, to study a different kind of air pollution: wood smoke.
In coastal African villages, women spend hours smoking freshly caught fish over open fires as a method of preserving the food without refrigeration. Bentil deployed the open-path sensor to measure water vapor and two organic molecules, 2-methyl phenol and benzyl alcohol, arising from the various species of burning firewood. Between the laser, retroreflector and detector, the beam traveled a total path length of 58 m. A commercial humidity gauge set up next to QCLOPS confirmed that the laser system was properly detecting water vapor. QCLOPS found the spectroscopic signatures of both aromatics as well as spectral lines from other organic gases, which Bentil was still working to identify at the time of her CLEO talk in 2011.
Zondlo and several colleagues revisited China in two month-long trips, June 2013 and June 2014, for an updated, more comprehensive look at the country’s air quality. They mounted their system on the roof rack of a passenger vehicle and logged roughly 16,000 km on each trip through Beijing and the North China Plain to measure ammonia, nitrous oxide, carbon monoxide and other pollutants. Measuring multiple gases at once lets scientists take a “chemical fingerprint” of the gases and try to figure out where they came from.
The group learned that vehicular emissions of ammonia are a significant source of urban air pollution in China and make a large contribution to the haze often captured in photos of that country’s large cities. Interestingly, Beijing’s vehicles give off ammonia at a rate similar to their counterparts in major American cities; Zondlo believes that’s because the two fleets are similar. Vehicles in rural, less developed regions of China do not have to meet the stricter emissions standards of the urban areas.
The Zondlo team’s second finding from the trips is less obvious. According to Zondlo, China has been encouraging the use of natural-gas vehicles to mitigate its air pollution problem because natural gas burns more cleanly than liquid petroleum products. A natural-gas-powered car can emit half the carbon dioxide of a conventionally fueled automobile.
Since natural gas primarily consists of methane, however, methane can unintentionally cause climatic harm if it leaks from fuel tanks or escapes from the tailpipe without fully combusting.
The group found that one city in China had much higher methane emissions than its counterparts in the United States and Europe. “If the Chinese government continues with their trend of trying to make 10 percent of the vehicle fleet natural-gas vehicles, China’s going to have a major source of methane coming from these cars if it doesn’t do something about these leaks,” Zondlo says.
The MIRTHE team has also hit the road in the United States to characterize air pollution with a mobile open-path sensor mounted on the roof rack of a passenger vehicle. This set of experiments featured a pair of CW DFB QCLs, one tuned to detect ammonia at 9.062 μm and the other set for carbon monoxide at 4.538 μm. Zondlo supplemented the lasers with a commercial CO2/H2O infrared sensor. Adding carbon monoxide and carbon dioxide levels together gives scientists an idea of contributions to pollution levels from vehicle fuel consumption.
One reason why open-path sensors are especially suited to trace-ammonia studies, according to Zondlo: Ammonia is “sticky.” Ammonia molecules somehow adhere to closed-cell walls and tubing walls made of Teflon, plastic or other materials generally considered unreactive. “So by the time you sample that air and get it to your sensor where it’s being analyzed, you’ve lost sometimes half of it [ammonia], sometimes 90 percent of it,” Zondlo says of the closed-path sensor systems. “It becomes really challenging to understand what the ammonia concentrations are, because you’re losing most of it on your instrument, or your instrument manifold.” With open-path sensors, the air flows freely through the two endpoints, giving the scientists high temporal resolution and more accurate ratios of the trace gases being studied.
After testing the open-path platform in the relatively controlled atmosphere of a parking garage, Zondlo’s group drove it around New Jersey (for 200 km) and California (for 4,300 km), the latter under the auspices of NASA’s four-year-long DISCOVER-AQ program for calibrating aircraft and satellite observations of air quality. Their West Coast travels took them down the busy U.S. Route 101 in Silicon Valley, across the Los Angeles basin and to three cities in the San Joaquin Valley. The Princeton team’s data confirmed some prior on-road emissions measurements but suggested that other emission studies may have underestimated vehicle emissions, especially in the northeastern United States.
 Aerial view of East Harlem apartment building explosion, New York, 12 March 2014. [Adnan Islam / Wikimedia Commons]
Aerial view of East Harlem apartment building explosion, New York, 12 March 2014. [Adnan Islam / Wikimedia Commons]
CCNY’s Moshary, who is also affiliated with MIRTHE, envisions that open-path QCL-based sensing could detect “hot spots” of methane emissions in urban environments. A pair of natural-gas explosions in Manhattan, each of which destroyed several buildings, underscored the urgency of the CCNY research. In March 2014, a blast in East Harlem leveled two apartment buildings, killed eight people and injured more than 70 others. One year and two weeks later, another explosion in the East Village burned three buildings to the ground, with two deaths and 19 injuries. Many natural-gas lines in New York and other cities are aging; one gas main near the 2014 explosion was originally installed in 1887.
Moshary’s Optical Remote Sensing Laboratory is adapting QCLOPS to find traces of methane by detecting backscatter from mid-infrared beams reflected from “targets of opportunity,” such as trees and buildings in the urban environment. The system currently picks up deliberately released puffs of natural gas; Moshary hopes that mobile open-path sensors can someday cruise the urban streets, looking for gas-line leaks before buildings go up in flames.
Looking to the future
This year, Zondlo has been driving around West Virginia and western Pennsylvania to study fugitive methane emissions from well pads in the Marcellus Shale, a natural-gas-rich sedimentary rock formation extending through much of the Appalachian Basin. According to the Princeton professor, industry officials claim that little methane escapes from shale drilling sites, though other studies suggest the opposite.
Zondlo wonders whether the region has a “fat-tail” problem, where a small percentage of well pads are giving off a large percentage of the leaks. He’s taking the mobile open-path sensor system, tuned to the spectral features of methane and ethane, to 1,000 different drilling sites to see the resulting patterns of emissions. (Ethane is not naturally found in Earth’s atmosphere, so it serves as a useful marker for petrochemical activity.)
QCL researchers are still increasing the power output and shrinking the packaging of mid-infrared laser diodes. “In the relative scheme of things these are very young lasers, and as they evolve and become more powerful and new variations come on the market, they make it possible to do different techniques,” Moshary says.
Zondlo credits MIRTHE with bringing academic researchers together with the “applications people” and industry representatives to advance the technology in the desired directions. He says, “Without that kind of constructive feedback, I don’t think mid-infrared lasers would be where they are today.” Zondlo adds that with open-path systems, “we can help understand air pollution better on a global continuous scale,” and that ongoing research “shows the impact that quantum cascade lasers are having on environmental issues basically throughout the globe.”
Patricia Daukantas is a freelance writer specializing in optics and photonics.
References and Resources
-
A.P.M Michel et al. Opt. Engin. 49(11), 111125 (2010).
-
E.N. Bentil et al. CLEO 2011, paper JMC6 (2011).
-
D.Q. Rich et al. JAMA 307, 2068 (2012).
-
L. Tao et al. Opt. Express 20, 28106 (2012).
-
D.J. Miller et al. CLEO 2013, paper ATh1I.4 (2013).
-
K. Sun et al. Environ. Sci. Technol. 48, 3943 (2014).
-
R.A. Rohde and R.A. Muller. PLoS ONE 10(8): e0135749 (2015).

